Welcome to Science with Shrike! After a long hiatus, we’re going to discuss a reader request: masks and efficacy. At this point, it’s far enough out from the pandemic that emotions are not as high on this topic as they once were. However, most have formed beliefs on the topic, generally due to the policy-makers. Here we will explore what science underlies masking. In part I, we will go through an overview of respiratory risks and masks. In part II, we’ll see how well they work.
Respiratory Risks
In order to understand the potential benefits of masks, we first need to discuss what they protect against. The main point of protection is from respiratory damage, be that infectious or particulate in nature. However, a secondary use is to reduce the risk of contaminating a surface (think surgeon working with an open wound, or a dentist in spitting distance of your open mouth). When it comes to respiratory risks, the composition of the particulate, and the size of the particulate are the two greatest factors for considering how bad it is.
For size, the smaller the particle, the more dangerous. This is because small particles stay airborne longer, and are able to penetrate deeper into the lungs. In the industrial world, the risks of particulates are well known. Sanding and spraying are two well-known risks, though other activities that generate dusts may also carry risks. The finer the dust or aerosol, the deeper it will make it into your lungs, and the longer it will stay airborne.
When it comes to human secretions, respiratory droplets are larger particles that fall to the ground reasonably quickly, whereas droplet nuclei are the small ones that stick around in the air longer. While people draw the lines at different sizes, respiratory droplets are generally 10 to 1000 μm, while droplet nuclei are ~5 μm or smaller. If you assume these droplets are spheres, this means respiratory droplets have 8-8,000,000 times the volume of each droplet nuclei.
When people exhale, they breathe out droplets of all sizes. Sneezing produces the most in one go (and can impart reasonable speed to the droplets), though coughing and singing also produce a lot. Talking gives fewer, and just breathing produces the fewest. The droplet nuclei will act as aerosols (liquids dispersed in a gas), and since they are small enough, will float around for a while. General estimates for aerosols to settle/turnover is ~15-20 minutes, though this is a rough ballpark. Note that a more concentrated aerosol would take longer to settle, and ventilation will change this a lot.
The other big factor for risks is the composition of the particulate. This is also well-known in the industrial world thanks to asbestos and coal mining. Certain particulates, like asbestos, cause problems due to their shape and chemical composition. Other particulates (like most dusts you breathe from mowing the lawn or if you end up in a dust storm) are well tolerated. While there are some adverse effects for some people, dust storms are more of a nuisance than outright health threat. Even with pollution, many people see no ill effects with short-term exposure. It takes chronic-exposure (and/or genetic, or other health pre-dispositions) to cause major problems. When it comes to chemical/solid hazards, the risks are well-studied, and well known. With a few exceptions, exposure amount and duration drives the pathology. In the US, OSHA sets standards for the acceptable level of exposure for a wide range of particulates that can be encountered during industrial or other commercial activity.
These ranges are not controversial, nor is the protection recommended for these particulates controversial.
When the composition of the particulate is a biologic, it becomes harder to assess. In part, this is because the occupational exposure at scale does not happen for biologics, and we didn’t figure out how to work with biologics until after most of the labor-protections for respiratory hazards were enacted. The closest occupational exposure for respiratory pathogens are health care workers dealing with patients who have one or more respiratory pathogens.
The challenge with infectious agents is that there are three outcomes that matter:
No apparent disease, and you cannot spread to other people
No apparent disease, but you can spread it to other people
Disease, which you can spread to other people.
While dose helps determine infection vs no infection, disease severity is not linear with dose. This stands in contrast to most particulate problems, where the more exposure you have, the more it makes your health problems worse. With infectious particles, it takes doses much higher than the infectious dose, before you observe dose-dependent effects on lethality/severity of outcome, with the caveat that the difference between the lethal dose 50% (dose needed to kill 50% of recipients) and infectious dose 50% (ID50, the dose needed to infect 50% of recipients) varies by pathogen. These are often S-shaped curves.
Autist note. S-shaped curves are logistic curves, modeled as f(x) = L/(1+ek(x-c))+D, where L is the saturation point, k is steepness, c is the value of x that is 50% between L and D, and D is any offset from zero. For our purposes, D is 0, L is 95-100 and c is the ID50).
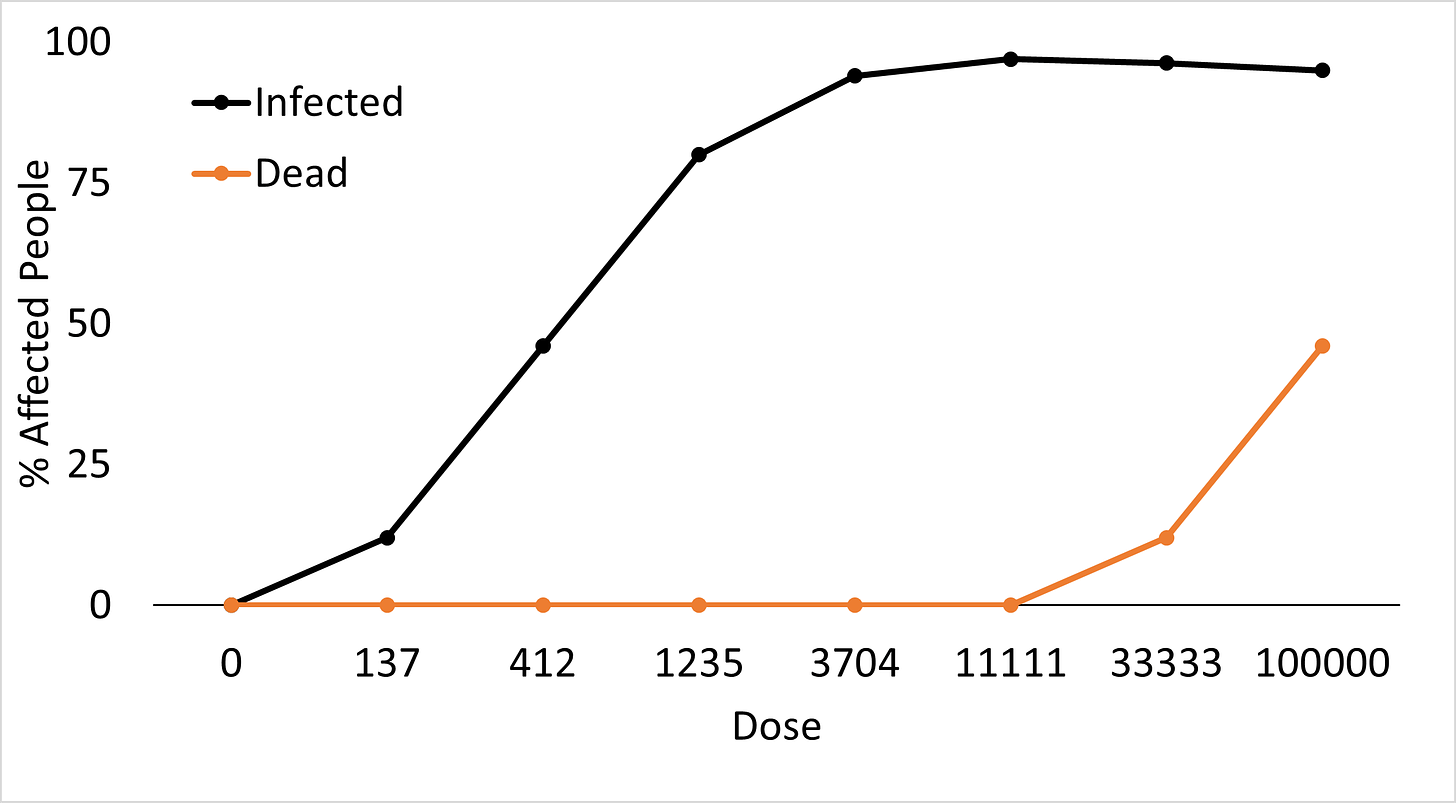
One key thing to note on S-shaped curves is that the curve changes the most around the 50% value. Around these values, a 2-3x change could go from 95% get infected to 5% get infected. However, above these values, even a 10x change might go from 95% get infected to 94% get infected. This stands in stark contrast to many particulates, where cutting exposure by 2-3x cuts risk 2-3x, and cutting exposure 10x may cut risk 10x for particulates. This means for infectious agents, it is important to know where the ID50 lies.
To summarize, for infectious particles, we need to know two things:
If they are spread mostly by large, respiratory droplets, or aerosolized in droplet nuclei, and
What the ID50 of the pathogen is. If the ID50 is low, a 2-3x reduction in particles may be meaningless because you remain above the ID50.
Types of Masks
Masks used for personal protection exist to counteract specific threats. Prior to 2020, most of these uses were well-established and not controversial. With the SARS-CoV2 pandemic, the prior rules about mask use for biological hazards went out the window. For example, prior to 2020 your employer could have been sued into oblivion for making you wear a cloth mask where a surgical mask was recommended, nevermind if an N95 was needed. Most of these rules changes came due to the scale of implementing the rules, and general lack of mask availability. Remember that rules are set by policy makers, and they may or may not reflect the science.
Surgical masks are designed to block respiratory droplets. This way the dentist does not spit in your mouth while working on your teeth. These masks are effective for their designed purpose—blocking liquid projectiles coming out of your mouth. But. They are not designed to stop aerosols. When you breathe out through the mask, all that air goes around the mask, often past your eyes or around your cheeks. Aerosols are carried on your breath, which keeps them from being trapped in the mask they flow around. For a fun exercise in ‘trapping aerosols’, try rescuing a small piece of egg shell that you accidentally got in your batter. A cushion of egg white will make recovering that piece of shell much harder. Same thing happens with aerosols in the air—they have an air cushion keeps them from sticking to the first thing they encounter.
Note that when you inhale, some air comes through the mask, but more of the air comes around the mask.
This is why OSHA forbids the use of surgical masks in place of respirators and other respiratory protection, and why no one uses surgical masks to protect from known aerosol-spread pathogens like Mycobacterium tuberculosis.
N95 are the standard entry-level respiratory protection for the user. They are intended to reduce particulate matter you breathe in by 95%. If vented, they do not do much to reduce aerosols or other materials you exhale. The KN95 is the Chinese knockoff of the N95 that does not fit as well. If you think you need respiratory protection, an N95 is where you started prior to 2020. In addition to needing to pass standards for the material construction, the masks also need to fit tightly against your face. This prevents the problem of air flow around the mask instead of through the mask. If the mask is NOT tightly fitted, it is not much better than a surgical mask.
N95s and other respirators require fit testing, aka making sure the device is working like it should be. This is an OSHA requirement for respirator use in the workplace. The simple way to do this is with a nebulizer and a bitterant, like Bitrex. You put the mask on, and then spray the bitterant around your face. If you can smell the bitterant, your mask is not making a tight fit, and you need to adjust it. Repeat until you can no longer smell the bitterant. Then test the mask when you breathe deep, move your head around, talk, bend over, smile and frown to ensure the mask will not get displaced in motion. Note that certain beard styles will prevent a tight fit for an N95.
Other versions of the test use saccharin (sweet), a banana smell (for filtering out volatile organic compounds), or an involuntary cough reflex. There are also more involved quantitative tests, but you are unlikely able to do those at home.
Respirators can get more complex. A P100 will filter >99.97% of particulates. Prior to 2020 these were used for hazardous particulates like mold spores and other occupational hazards. They protect your respiratory system, but not your eyes. They can be supplemented with goggles to cover your eyes.
Gas masks will also cover the eyes and most of the face, with rubber or silicone to help ensure the fit. This is what you wear for asbestos removal, as well as resisting toxic gasses and other chemicals. Activated charcoal is used to filter out the worst of the gas, allowing you to breathe ok. While activated charcoal can work well for chemical hazards, it is not intended for most biologic hazards.
If you want to fully shield your head, or can’t get a tight fit with a P100, you can upgrade to a Powered Air Purifying Respirator (PAPR). This is a battery powered device that pumps air through a series of filters, and delivers it to your face. You will need the fancier systems if you want it to be “intrinsically safe” and to provide both HEPA and chemical filtration. You also wear something to enclose your head, protecting you from the outside environment. These can be reusable or disposable. It is important when looking at these to select one that is rated for what you want. While the PAPR reduces the need for perfect fits, it is only as good as the filters through which it pumps air.
For biological hazards that require the highest biosafety containment (BSL-4, for pathogens like Ebola), you need the full “space suit”. This is a positive pressure suit that fully encloses the body with clean air supplied to the suit. Since BSL-4 labs are few and far between, these will be rare to encounter. Generally at the consumer/ industry level, problems for which you might want a BSL-4 suit may be handled either by pairing a Tyvek or HazMat suit with one of the above respirators, gloves and goggles, or by fixing the air supply. Ventilation is a critical, but separate issue.
With all of these solutions, there are also practical considerations:
What is your range of vision?
Does it fog or limit your vision?
How well does it deliver air to your lungs?
Does it physically interfere with anything you are doing?
How well can you tolerate wearing it for the full duration of your task?
Source Control?
Most of these devices are designed to protect the wearer, not the environment. Purifying the air as it leaves from a potentially infectious person is not practical for several reasons. First, it is hard to determine who is infectious and who is not. Second, many of the filtration solutions vent the air back into the environment, so specialized protection is needed. Last, in the source control scenario, your safety depends on another person’s compliance instead of yours.
To detoxify the environment, ventilation is a more robust solution. Note that places with particulates as an occupational hazard encourage both good ventilation paired with an appropriate respirator.
In part II, we will dig into how masks can be evaluated for infectious particles, both in the lab and in clinical trials.

